Your Random Science News Story From the Month of May
News outlets reporting on the latest health research often underplay (or flat out ignore) the fact that a lot of the research involved animals outside of their readership demographic – you know, species wise. So, we can see a lot of gun-jumping when headlines flash about the next best cancer treatment or drug to fight dementia (and so on and so forth), while human trials are still years if not a decade away. And the positive results displayed in, say, mice, are often not observed in people. Some human diseases/conditions are better modeled by animals than others, and whether or not animal experiments are all that valuable for predicting the efficacy of a drug in human trials remains controversial, for various reasons.
But sometimes the research being done just sounds too cool to leave in the pages of an academic journal, like a MIT study from late 2016 that claimed a strobe light could help battle Alzheimer’s. Or a story from last month where researchers from Shanghai designed a “sponge” that could regulate glucose levels in diabetic patients. I think these kinds of stories are fine to report on as long as you emphasize all the caveats that come with them.

The paper I’m about to cover is currently accessible in “Just Accepted” form from the Journal of the American Chemical Society, meaning that it’s been peer-reviewed but hasn’t completed all editing/formatting stages. It’s also behind a paywall, so you can’t read the whole thing unless you cough up the cash.
What happened?
Researchers Yufen Xiao, Hui Sun, and Jianzhong Du of Tongji University have designed a “sugar sponge”, as they call it, to be used as an alternative treatment to insulin and other anti-diabetic drugs. This sponge, of course, is neither your typical kitchen cleaner nor the simplest animal on the tree of life.
It’s nanotechnology, measuring a mere 390 nanometers in diameter, on average – roughly 200-250 times thinner than a human hair! These tiny “polymer vesicles”1 have been constructed to store extra glucose when there’s too much in the bloodstream, and release the stored molecules when the blood sugar level drops.
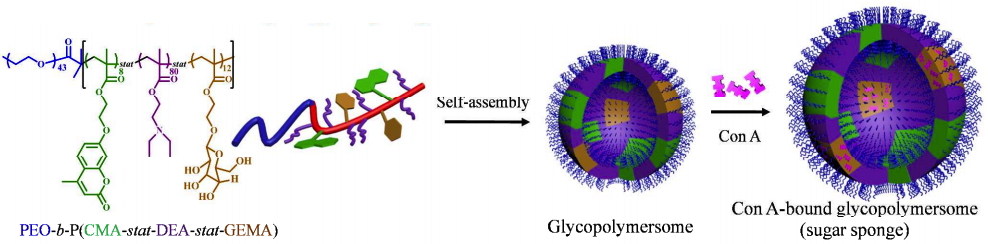
To make a “sugar sponge”, the researchers constructed the standard polymer vesicle (aka, as above, “glycopolymersome”2) then embedded in the membrane a special kind of protein that binds to carbohydrates, called a lectin. Different lectins have different affinities for different carbohydrate molecules, so they chose one that binds with glucose: Concanavalin A (aka Con Air). Con A prefers the glucose floating around in your blood to the glucose already built into the vesicle, so it binds to the vesicle when there’s no sugar needing soaking up, but when the time comes it swaps that for a free-floating glucose, effectively storing it for later.
But you might be wondering: How is it only the extra glucoses get soaked up? To answer that, we look to the simple scientific principle of diffusion. If the concentration of glucose molecules is higher on the outside of a vesicle than it is on the inside, then some of the molecules will diffuse through the membrane to get to the inside and balance the concentrations (If the concentration on the inside exceeds that of the outside, they move the other way). Upon this diffusion into the membrane, the glucose molecules bind with the Con A molecules embedded in said membrane – this causes the whole thing to swell up.3
After construction, the sugar sponges were tested in vitro (meaning outside a living organism) to observe if they could successfully both soak up glucose at higher concentrations, and release glucose at lower concentrations. (They were also tested for how they responded to the sugar mannose, which Con A can also bind to, but it’s much less abundant in the blood stream – glucose outnumbers it 100 to 1 – and doesn’t stick around long, either).
Then comes the in vivo (meaning inside a living organism) testing. The researchers constructed a “type I diabetic mice model” by injecting 31 8-week-old male Kunming mice with a compound that’s toxic to insulin-producing cells (it’s called “Streptozotocin”). After one week, 74% of them reached blood glucose levels high enough to be considered “diabetic”. The mice were either injected with the “sugar sponges”, sponges without the Con A, just a solution of Con A molecules, or a buffer solution for a control (A group of non-diabetic mice were given the control as well). Then, their blood was tested at 2, 6, 12, 24, 26, 48, and 75 hours post-injection.
What’d they find?
After constructing the sponges, they tested their “breathing behavior” against some control vesicles that didn’t have any Con A embedded in their membranes. As the term “breathing” implies, the Con A sponges were not only able to demonstrate that the sponges
- grew when placed into a solution of glucose with a higher concentration than they had previously been exposed to, and
- shrunk when exposed to a lower concentration solution,
they could also oscillate in size if the concentration changed back and forth. (Specifically, the concentrations were 0.5 and 1.25 mg of glucose per mL of water, and they bounced between 500ish nm and 650ish nm in diameter at least four times). The controls, as expected, did not vary in size.
As for the mice models,
- Mice given the buffer and the non-Con A sponges showed little to no change in overall blood glucose levels (BGL) over the testing period.
- Mice given the Con A died after 36 hours because, as it turns out, the protein all by itself (that is – not embedded in the sponge’s membrane) is toxic to cells. Before that, their BGL dropped to healthy levels 2 hours post-injection but jumped back up by the 6-hour mark.
- Only the “sugar sponge” mice had their BGL drop to non-diabetic levels and then slowly rise back to their original levels over the course of the 3 days.
The researchers note that their sponges function similar to a molecule we already have in our body, called “glycogen” (which doesn’t look anything like the fluid-filled spheres we’re talking about, here…).
So what?
Diabetes is a pandemic. In 2010, it was the 7th leading cause of death in the United States. The American Diabetes Association estimates that the total cost of treating diabetes (and prediabetes) is 322 billion dollars, with approximately 1 in 11 Americans currently suffering from the condition. According to the CDC, by 2050 that might rise all the way to 1 in 3. So, it’s safe to say, finding better treatments is an important area of research, right now.4
Keeping in mind that this study was done on mice – and therefore isn’t directly translatable to humans – it shows that there may be a viable alternative treatment for diabetes (both type I and II) that doesn’t require insulin or any other kind of medication. If the results of future human trials come out similar to those of this study – specifically that the “sugar sponges” were able to maintain ‘normal’ BGL for roughly two days with one dose – the sponges may prove a more efficient treatment than the much shorter-lasting insulin injections, and one that comes with fewer negative side effects.
One possible point of worry is that the sponge membranes contain a toxic component called coumarin (about 10% by weight); the researchers note that these components are buried inside the membrane and thus don’t make contact with any body cells, and the level of toxicity tested on a particular type of liver cell was low. They didn’t make note of any health complications in the mice given the “sugar sponges” or the non-Con A glycopolymersomes, but they may not have been observed for long enough for any side effects to start showing.
It’s also important to note the very small sample size of mice used in this study – only 5 mice were given the “sugar sponges” (or any other injection option), which is no where near enough data points for us to start testing in humans.
But, as is, I think this is a very good preliminary study working on a nanoscale solution to a very big problem.
1. Polymer = a large molecule made up of repeating subunits, Vesicle = a small cavity. The polymer in this case was made from “poly(ethylene oxide)-block-poly[(7-(2-methacryloyloxyethoxy)-4-methylcoumarin)-stat-2-(diethylamino)ethyl methacrylate-stat-(α-D-glucopyranosyl)ethyl methacrylate]”, a name that means absolutely nothing to me. ↩
2. You might be wondering how they got the glycopolymersome in the first place – they made that, too, but it self-assembled into a hollow sphere because parts of the polymer’s subunits are hydrophobic and parts are hydrophilic – just like the membranes to your own body cells do.↩
3. It’s basically like how you make room for a new person joining your conversation circle by making it bigger – not forcing the new-comers to stand awkwardly in the middle of the (originally-sized) circle. ↩
4. Yes, funding projects/policy that would lead to preventing diabetes in the first place is important, too. That’s not what this news story is about. ↩
